Glass
In this section you will find information on glass composition, and information on the manufacturing process of various types of glass and how it relates to the glass appearance.
Glass Composition
|
Ingredients & Properties.
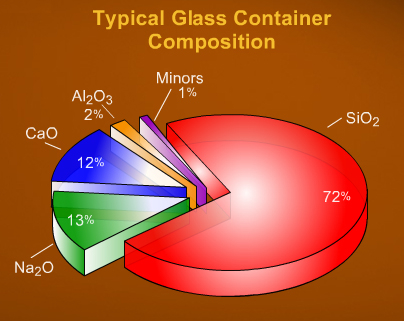
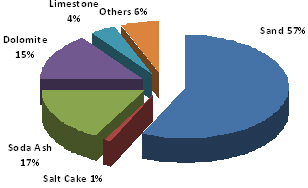
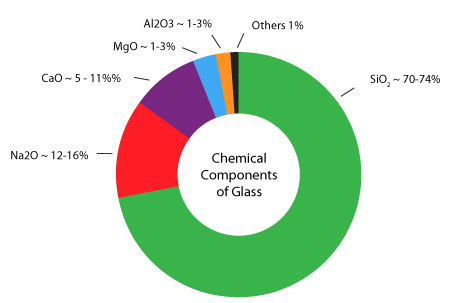
Raw materials: Silica (~ 60 – 70 %) Lime (~ 13 %) Soda (~ 12 %) A batch consists of 75 % raw materials and 25 % recycled waste glass (cullet). A variety of other materials may be added to the batch to change characteristics like color, brilliance, density, expansion coefficient, etc. Proportions and ingredients vary with the type of glass, as illustrated by the charts to the right. Silica (sand; silicon dioxide) fuses into glass at 3133 degrees Fahrenheit. Soda ash (sodium carbonate) or caustic soda (sodium hydroxide) lowers the melting point to 2600 – 2900 degrees Fahrenheit, but the resulting glass is water-soluble. Lime (calcium oxide) makes the water glass insoluble. The addition of cullet (waste glass) further lowers the temperature required to fuse the ingredients. Glass is an amorphous (non-crystalline) solid material. It is both brittle and optically transparent. Technically the term glass applies to any non-crystalline solid substance which transitions through a glass phase as it is heated towards its melting temperature. While the term glass applies to such materials as metallic alloys, aqueous solutions, molecular liquids, and polymers, it most commonly refers to the specific soda-lime glass used for windows and drinking vessels. |
|